Quick Details
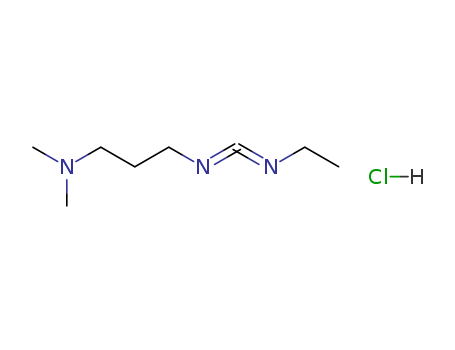
- ProName: 1-(3-Dimethylaminopropyl)-3-ethylcarbo...
- CasNo: 25952-53-8
- Molecular Formula: C8H18ClN3
- LimitNum: 0
Superiority
Molecular formula: C8H17N3.HCl
Molecular weight: 191.7
Shape: white powder
IR spectrum: qualified
Assay:≥98%
Solubility: clear colorless solution (1g/ml,H2O)
Uses: dehydrating agent of poly…
Details
Molecular formula: C8H17N3.HCl
Molecular weight: 191.7
Shape: white powder
IR spectrum: qualified
Assay:≥98%
Solubility: clear colorless solution (1g/ml,H2O)
Uses: dehydrating agent of polypeptide , protein , nucleotide
Storage temp:2-8°C
Related information of EDC.HCl
1-Ethyl-3-[3-dimethylaminopropyl]carbodiimide hydrochloride is a zero-length crosslinking agent used to couple carboxyl groups to primary amines. This crosslinker has been used in diverse applications such as forming amide bonds in peptide synthesis, attaching haptens to carrier proteins to form immunogens, labeling nucleic acids through 5’ phosphate groups and creating amine-reactive NHS-esters of biomolecules. 1-ethyl–3-(3-dimethylaminopropyl) carbodiimide hydrochloride reacts with a carboxyl to form an amine-reactive O-acylisourea intermediate. If this intermediate does not encounter an amine, it will hydrolyze and regenerate the carboxyl group. In the presence of N-hydroxysulfosuccinimide (Sulfo-NHS), EDC.HCl can be used to convert carboxyl groups to amine-reactive Sulfo-NHS esters. This is accomplished by mixing the EDC.HCl with a carboxyl containing molecule and adding Sulfo-NHS.
EDC.HCl reacts with a carboxyl group on molecule #1, forming an amine-reactive O-acylisourea intermediate. This intermediate may react with an amine on molecule #2, yielding a conjugate of the two molecules joined by a stable amide bond. However, the intermediate is also susceptible to hydrolysis, making it unstable and short-lived in aqueous solution. The addition of Sulfo-NHS (5 mM) stabilizes the amine-reactive intermediate by converting it to an amine-reactive Sulfo-NHS ester, thus increasing the efficiency of EDC.HCl -mediated coupling reactions1,2. The amine-reactive Sulfo-NHS ester intermediate has sufficient stability to permit two-step crosslinking procedures, which allows the carboxyl groups on one protein to remain unaltered.
1-Ethyl-3-[3-dimethylaminopropyl]carbodiimide hydrochloride Highlights
Efficiency of EDC.HCl -mediated coupling is increased in the presence of Sulfo-NHS
By combining EDC.HCl and Sulfo-NHS, amine reactive Sulfo-NHS esters can be created on any carboxyl-containing molecule
Amide bonds formed with this crosslinking reaction provide a neutral linkage
Excess reagent and crosslinking byproducts are easily removed by washing with water or dilute acid
EDC.HCl is water soluble, so crosslinking can be done in physiologic solutions without adding organic solvent
High purity, crystalline EDC.HCl can be used to create high-purity activated derivatives
Applications for 1-Ethyl-3-[3-dimethylaminopropyl]carbodiimide hydrochloride
Conjugate carboxyl to amine groups in peptides and proteins
Convert carboxyls to amine-reactive Sulfo-NHS esters
Crosslink proteins to carboxyl coated beads or surfaces
Activate nanoparticles with amine-reactive Sulfo-NHS esters
Couple haptens to carrier proteins (e.g. attach a peptide to KLH)
DNA labeling through 5’ phosphate groups